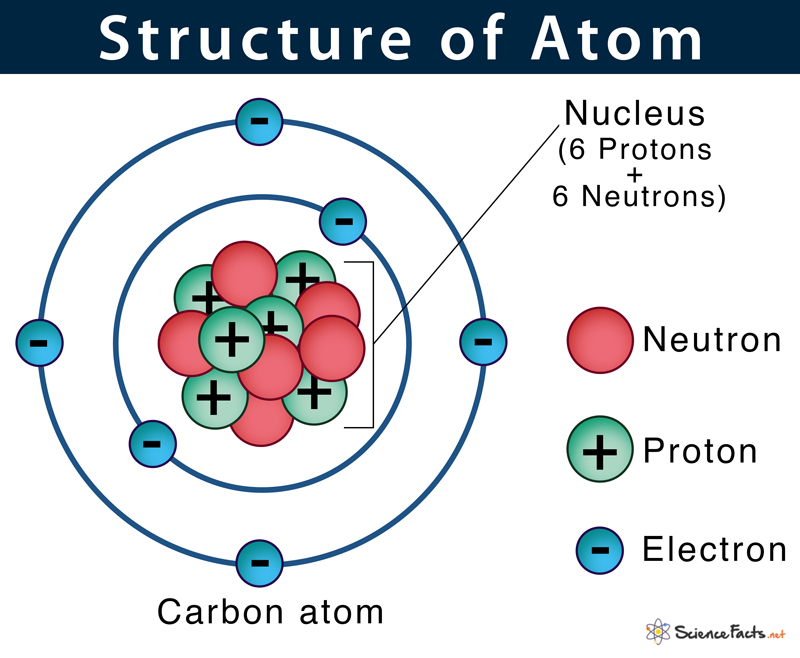

The complete number of protons is equivalent to the number of electrons rotating around the nucleus, and henceforth the atom, in general, is electrically neutral. Nuclear energy depends on only discharging the energy caught in the nucleus of an atom. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic forces of repulsion likewise act inside the nucleus. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. Henceforth, on the whole, the nucleus of an atom is positively charged, and the electrons which are negatively charged rotate around the center of the nucleus.
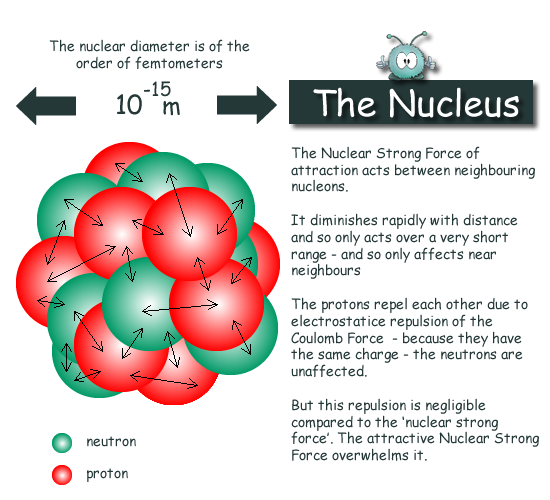
Of the two, the protons have a net positive charge. These are the two solid particles in an atom, and consequently, 99.9% of the mass is gathered in the nucleus. The nucleus of an atom consists of proper and tightly arrangement of protons and neutrons. The mass of a hydrogen atom can be expressed as follows:ġ u = one particle of C-12/12 = 1.992647 10 -26 /12 kg Its reference is taken as Carbon-12, and 1 Atomic Mass unit is equivalent to 1/twelfth the weight of one atom of Carbon 12. This is termed as Atomic Mass Unit with symbol u. An ordinary unit of mass, such as a Kilogram (Kg), can't be utilized to gauge something as small as an atom, and to deal with this agenda, researchers have made another unit of mass. The nucleus of an atom is little to the point that if you extended an atom to occupy a room, the nucleus of an atom would at present be no bigger than a pinhead!Īn Atom is tiny, and in this manner, its mass is additionally relatively minute. Numerically, the nucleus of an atom possesses almost nearly 10-14 times the volume of the atom yet contains 99.99% of the atomic mass. Through the dispersing of alpha particles explored by Rutherford, we discovered that the nucleus of an atom contains a more significant part of the mass of the atom. An ion with extra electrons is negatively charged and is called an anion an ion deficient in electrons is positively charged and is called a cation.The nucleus of an atom is the focal locale of an atom where most of the mass is concentrated. An ion is an atomwith extra electrons or with a deficiency of electrons, resulting in itsbeing electrically charged. Protons and electrons have equal and opposite charge, and normally anatom has equal numbers of both. Examples of different isotopes of an element are carbon 12(the most common, non-radioactive isotope of carbon) and carbon 14 (a less common, radioactive isotope of carbon). Theisotope for an element is specified by the sum of the number of protons andneutrons. Because the number ofneutrons in an atom can vary, there can be several different atomic weights for mostelements.Ītoms having the same number of protons, but different numbers ofneutrons, represent the same element, but are known as different isotopes of that element.
#THE NUCLEUS OF AN ATOM CONTAINS PLUS#
For most practical purposes, the atomic weight can be thought of as the number of protons plus the number of neutrons. Electrons contribute only a tiny part of this mass. The total mass of an atom, including the protons, neutrons andelectrons, is the atomic mass or atomic weight. When it loses a certain amount of energy, it falls to a smaller, or lower, shell. When an electron absorbs enough energy, it moves to a larger, or higher, shell. These distances are represented by spheres, called shells, surrounding the nucleus. In the Bohr atom,the negatively charged electrons orbit the nucleus at specific mediandistances. Niels Bohr revised Rutherford's theory in 1913. In the so-called Rutherford atom, electrons orbit the nucleus in circular paths. He was the first to suggest that atoms are like miniature solar systems, except that the attractive force is not caused by gravity, but by opposing electrical charges.

The mass of a proton or neutron increases when the particle attains extreme speed, for example in a cyclotron or linear accelerator.Īn early model of the atom was developed by the physicist ErnestRutherford in 1912. A neutron is electrically neutral and has a rest mass, denoted m n, of approximately 1.675 x 10 -27 kg. A proton has a rest mass, denoted m p, of approximately 1.673 x 10 -27 kilogram (kg). The number of protons in the nucleus ofan atom is the atomic number for the chemical element. The nucleus is positively charged, and contains one or more relatively heavy particles known as protons and neutrons.Ī proton is positively charged. An atom consists of a central nucleus that is usually surrounded by one or more electrons. An atom is a particle of matter that uniquely defines achemical element.
